PHARMACEUTICAL WATER
A) Introduction
Water is widely used as a raw material, ingredient, and solvent in the processing, formulation, and manufacture of pharmaceutical products, active pharmaceutical ingredients (APIs) and intermediates, compendial articles, and analytical reagents. This general information provides additional information about water, its quality attributes that are not included within a water monograph, processing techniques that can be used to improve water quality, and a description of minimum water quality standards that should be considered when selecting a water source.
There are many different grades of water used for pharmaceutical purposes. Several are described in USP monographs that specify uses, acceptable methods of preparation, and quality attributes.
These waters can be divided into two general types:
- Bulk waters, which are typically produced on site where they are used.
- Packaged waters, which are produced, packaged, and sterilized to preserve microbial quality throughout their packaged shelf life.
There are also other types of water for which there are no monographs. These are all bulk waters, with names given for descriptive purposes only. Many of these waters are used in specific analytical methods. These nonmonographed waters may not necessarily adhere strictly to the stated or implied modes of preparation or attributes.
B)Types of Water Used:
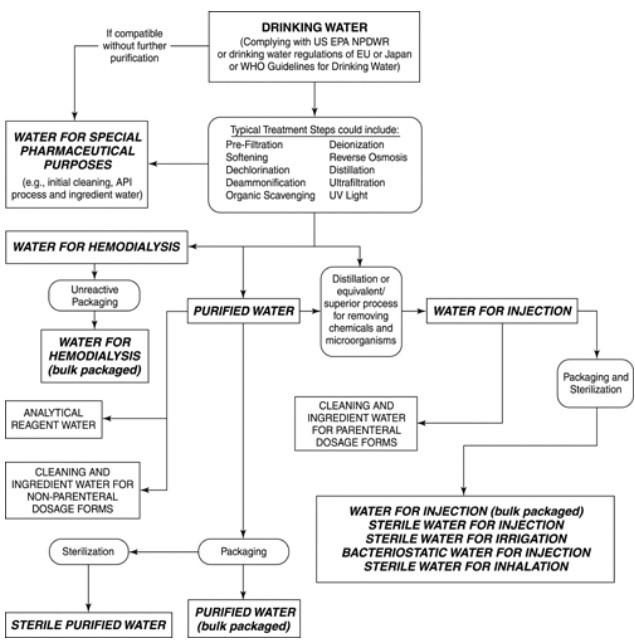
-
- Non potable water: It is not of drinking water quality, but may still be used for many other purposes, depending on its quality. Non-potable water is generally all raw water that is untreated, such as that from lakes, rivers, ground water, springs and ground wells.
Purposes:
cleaning of outer surface of the factory
used in garden
washing vehicles etc- Portable water: It is not suitable for general pharmaceutical use because of the considerable amount of dissolved solids (chlorides, sulphates and bicarbonates of Na, K, Ca and Mg present. A 100 ml portion of official water contains not more than 100 mg of residue (0.1%) after evaporation to dryness on a steam bath.
Purposes:
To use as drinking water
Washing and the extraction of crude drugs
Preparation of products for external use- Purified water: Purified water is used in the preparation of all medication containing water except ampoules, injections, some official external preparations such as liniments. Purified Water must meet the requirements for ionic and organic chemical purity and must be protected from microbial contamination. The minimal quality of source or feed water for the production of Purified Water is Drinking Water.
Purposes:
For the Production of non-parenteral preparation/formulation
For the Cleaning of certain equipment used in non-parenteral product preparation
For Cleaning of non-parenteral product-contact components
For All types of tests & assay
For the Preparation of some bulk chemicals
For the preparation of media in microbiology laboratories
Preparation technique:- Deionisation
- Distillation
- Ion exchange
- Reverse osmosis
- Filtration etc.
- Water For Injection (WFI): It is used in the production of parenteral and other preparations where product endotoxin content must be controlled, and in other pharmaceutical applications. It is sterile, non-pyrogenic, distilled water for the preparation of products for parenteral use. It contains no added substance and meets all the requirements of the tests for purified water. It must meet the requirements of the pyrogen test. The finished water must meet all of the chemical requirements for Purified Water as well as an additional bacterial endotoxin specification. Since endotoxins are produced by the kinds of microorganisms that are prone to inhabit water, the equipment and procedures used by the system to purify, store, and distribute Water for Injection must be designed to minimize or prevent microbial contamination as well as remove incoming endotoxins from the starting water.
Purposes:
For the production of parenteral products/formulation
For cleaning of parenteral product-contact components
Preparation technique:
Distillation
Reverse osmosis
Membrane process
Storage condition:
It can be stored for periods up to a month in special tanks containing ultraviolet lamps. When this freshly prepared water is stored and sterilized in hermitically sealed containers, it will remain in good condition indefinitely.
If autoclave is not available, freshly distilled water may be sterilized by boiling the water for at least 60 minutes in a flask stoppered with a plug of purified nonabsorbent cotton covered with gauze, tin-foil or stout nonabsorbent paper; or the neck of the flask may be covered with cellophane and tightly fastened with cord.- Sterile water for injection: It’s specifications are provided in USP monograph for water for injection, sterilized and packaged in suitable single-dose containers, preferably of type I glass, of not larger than 1000 ml size. It meets the requirements of the sterility test and pyrogen test and other tests under purified water.
Purposes:
Used for extemporaneous preparation compounding
Used as a sterile diluent for parenteral products
Preparation technique:
By distillation of water for injection (WFI)- Bacteriostatic WFI: This is sterile Water for Injection containing bacteriostatic (antimicrobial) agents. It may be packed in single-dose containers of not larger than 5 ml size and in multiple-dose containers of not larger than 30 ml size, the label of which indicates the name and the proportion of added agent.
Purposes:
Used as a diluent in the preparation of parenteral products
Preparation technique:
By using sterile water for injection- Sterile water for Inhalation: Sterile water for Inhalation is Water for Injection that is packaged and rendered sterile and is intended for use in inhalators and in the preparation of inhalation solutions. It carries a less stringent specification for bacterial endotoxins than Sterile Water for Injection, and therefore, is not suitable for parenteral applications.
Purposes:
Preparation of use in inhalators
Preparation of inhalant solutions
Preparation technique:
By sterilization of water for injection- Sterile water for irrigations: Sterile water for irrigations is Water for Injection packaged and sterilized in single-dose containers of larger than 1 L in size that allows rapid delivery of its contents. It need not meet the requirement under small-volume injections.
Purposes:
To bath and moisten body tissue
Performing urologic procedure for surgeon
Preparation technique:
From water for injection
Example:
Surgical irrigation solution (Splash solution)
Urologic irrigation solution
Glycine solution
Sorbitol solution- Water for hemodialysis: Water for hemodialysis is used for hemodialysis applications. It may be packaged and stored in unreactive containers that preclude bacterial entry. The term “unreactive containers” implies that the container, especially its water contact surfaces, are not changed in any way by the water, such as by leaching of container-related compounds into the water or by any chemical reaction or corrosion caused by the water. The water contains no added antimicrobials and is not intended for injection.
Purposes:
For the dilution of hemodialysis concentrate solution
Preparation technique:
From safe drinking water- Pure steam: Pure steam is also sometimes referred to as “clean steam”.
Purposes:
To remove any co-deposited impurity residues
For air humidification in controlled manufacturing environments
Used in steam sterilization of equipment and porous loads
For cleaning the places where condensate directly comes in contact with official articles, product contact containers, and surfaces.
Water miscible solvents: Although water miscible solvents are used in parenteral, principally to enhance drug solubility, it is important to mention that they also serve as stabilizers for those drugs that degrade by hydrolysis. A water miscible solvent must be selected with grade care for it must not be irritating, toxic, or sensitizing, and it must not exert an adverse effect on the ingredients of the formulation. Solvents that are miscible with water are:
dioxolanes
dimethylacetamide
butylene glycol
polyethylene glycol 400 and 600
propylene glycol
glycerin and
ethyl alcohol
Water immiscible solvents include:
Fixed oil
ethyl oleate
Isopropyl myristate, and
Benzyl benzoate
C) NON-monographed Water -
- High Purity Water: It is water that is prepared by deionising previously distilled water, and then filtering it through a 0.45-µm rated membrane. This water must have an in-line conductivity of not greater than 0.15 µS/cm (6.67 Megaohm-cm) at 25.
- Ammonia-Free Water: Functionally, this water must have a negligible ammonia concentration to avoid interference in tests sensitive to ammonia. It has been equated with High Purity Waterthat has a significantly tighter Stage 1 conductivity specification than Purified Water because of the latter’s allowance for a minimal level of ammonium among other ions.
- Carbon Dioxide-Free Water: The introductory portion of the Reagents, Indicators, and Solutions section defines this water as Purified Waterthat has been vigorously boiled for at least 5 minutes, then cooled and protected from absorption of atmospheric carbon dioxide. Because the absorption of carbon dioxide tends to drive down the water pH, most of the uses of Carbon Dioxide-Free Water are either associated as a solvent in pH-related or pH- sensitive determinations or as a solvent in carbonate-sensitive reagents or determinations. Another use of this water is for certain optical rotation and colour and clarity of solution tests.
- Deaerated Water: This water is Purified Water that has been treated to reduce the content of dissolved air by “suitable means”. In the Reagents section, approaches for boiling, cooling (similar to Carbon Dioxide-Free Water but without the atmospheric carbon dioxide protection), and sonication are given as applicable for test uses other than dissolution and drug release testing.
Deaerated Water is used in both dissolution testing as well as liquid chromatography applications where outgassing could either interfere with the analysis itself or cause erroneous results due to inaccurate volumetric withdrawals.
FIND MORE AT…
Reference links
http://www.pharmacopeia.cn/v29240/usp29nf24s0_c1231.html
http://www.who.int/medicines/areas/quality_safety/quality_assurance/GMPWatePharmaceuticalUseTRS970Annex2.pdf?ua=1

Responses